
Most of the time, organizations like Media Matters are a bit too left-wing for my tastes. However, recently they blogged [1] about a Fox News segment where a meteorologist [2] claimed that one of the laws of thermodynamics “proves” that global warming cannot happen. The stark scientific ignorance illustrated by this Fox News segment got me thinking about the physics of the greenhouse effect. I figured it was time for a refresher. After all, the Greenhouse Effect is real, it happens, and is not in conflict with any law of nature (in fact, it’s an inevitable consequence of physics and chemistry) [3].
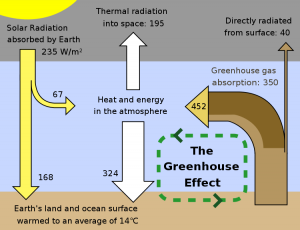
Quite simply, the greenhouse effect occurs when the temperature of a system rises to a higher level than that expected simply due to the action of an external input of energy. For instance, if the sun were the only way to heat the surface of the earth, our planet would be much cooler than it is (independent of the actions of our species) and relatively inhospitable to life as we know it. Gases in our atmosphere absorb the heat energy radiated by the surface of the earth and re-radiate it in all directions, including right back at the surface. This causes the surface of the earth to be much hotter than is possible only by the activity of the sun.
Let’s explore some of these ideas in a bit more depth. First, what is temperature? Temperature is a measure of the heat energy possessed by a system. The system can be made of many parts – for instance, air is made from an extremely large number of molecules (about 6.02×1023 in every 24 liters of air). Each molecule possesses a certain amount of energy, in the form of motion (translation), vibration, rotation, or combinations of all three. While each molecule may possess differing degrees of these fundamental energies, they collide with other molecules and exchange energy. This would seem an unfathomably complex set of behaviors – translation, vibration, rotation, collision with an unknown number of neighbor molecules, and exchange of energy during collision. Heat energy represents a statistical average of the behavior of all of these individual parts, and is a study all of its own (Statistical Mechanics, or “Stat Mech”). Temperature is a measure of the heat energy possessed by a system. [4]
When two systems are brought into contact, such that they can exchange energy (we’ll explore what that means in a bit), what determines how energy will be exchanged is the relative temperatures of the two systems. If one system possesses a higher temperature than the other, then heat energy will be transferred from the hotter system to the cooler system, until they reach a new and common temperature and no longer exchange heat energy. This is what is meant by “achieving thermal equilibrium” or reaching an “equilibrium state”. Two systems can begin each in isolation, unable to release or receive energy; in that case, they will maintain their original temperatures and each is said to be in their own equilibrium states. When brought into thermal contact, such that they can exchange heat energy, the new system they represent – the sum of the two – is NOT in its thermal equilibrium state. Energy must be exchanged until a common temperature is reached. This represents motion toward and achievement of equilibrium.
All of these ideas are summarized neatly in the laws of thermodynamics [5]:
- The Zeroth Law: if two systems, labeled A and B, are both in thermal equilibrium with a third system, labeled C, then A and B are in thermal equilibrium with each other.
- The First Law: the change in total energy of a system is the result of the heat energy added TO the system and the work done BY the system.
- The Second Law: a process which only transfers heat energy from a cooler system to a hotter system is impossible.
- The Third Law: in a perfect crystal (a regularly ordered solid) the entropy (a measure of disorder in a system) approaches zero as the temperature approaches zero.
Let’s close out this little post by exploring these laws and their relevance to the greenhouse effect. We’ve already explored the zeroth law a bit by thinking about a hot system coming into thermal contact with a cold system, and the new total system seeks a new equilibrium. Imagine that we have three such systems: A, B, and C. We bring A into contact with C and find no change in temperature; they must have been in thermal equilibrium already. We bring B into contact with C and also find no change in temperature, so they too are already in thermal equilibrium. Therefore, we know automatically that A and B are also in thermal equilibrium.
The first law tells us how the total energy of a system can be changed. If we add heat energy to that system, its total energy increases. If we do mechanical work on that system, its total energy also increases. If the system does mechanical work, its total energy decreases. If the system loses heat energy, as by coming into contact with a cooler system, it also decreases its total energy. The first law tells us how heat energy and work energy relate to changes in the total energy of the system, and is a restatement of energy conservation (in order to change you total energy, heat energy must change hands and/or work energy much change hands. If you lose, something else gains, and vice versa.
The second law places limits on the kinds of processes that can occur and helps define a “reversible” process (once whose effects can be reversed) and an “irreversible” process (one whose effects cannot be reversed without an external forcing factor). For instance, the law states that if two bodies are brought into contact and one is hotter than the other, it is IMPOSSIBLE for the hotter object to gain heat energy from the cooler object and thus increase the temperature difference between them. That is, it’s IMPOSSIBLE if the ONLY way energy can be exchanged is via heat energy. If there is also mechanical work being done, then it IS possible to transfer heat energy from the cooler object to the warmer one, as detailed in the first law. You refrigerator is just such a system. A mechanical pump does work on a fluid, and the fluid take heat energy from the inside of the fridge (system A) to the outside of the fridge (system B). System B is hotter than system A, but as long as the mechanical pump continues to do work (thanks to external energy supplied by the electrical grid), heat can move from cooler to hotter; when the pump fails or is switched off, heat can no longer make that journey as heat energy exchange then becomes the sole mechanism by which heat energy can be moved.
The third law tells us what happens to the disorder, or entropy, or a perfect crystal when that system is cooled (e.g. by doing work to remove heat energy from the crystal) toward absolute zero temperature. The disorder will tend to zero as well.
The earth is a system, but it is composed of many separate subsystems, and it is not isolated from external factors. All of the earth subsystems are at different temperatures, and thus are exchanging energy with one another and trying to reach a thermal equilibrium. Energy is injected into the earth primarily via the sun (some energy at the surface results from geothermal processes inside the earth). The energy causes the temperature of the surface to change, as well as that of the atmosphere. The surface radiates solar energy as heat and light; the atmosphere absorbs that and then it, too, re-radiates the energy (primarily as heat). The surface and atmosphere are in thermal contact, and their different temperatures promotes a constant exchange of energies that represents these systems trying to achieve thermal equilibrium. What is key in all of this is that the earth is not a closed and isolated system; it is in thermal contact with the sun, in that energy radiated by the sun can enter the earth system and affect the temperature of its various subsystems. So there is not only nothing here that violates laws of nature; these behaviors are all an INEVITABLE CONSEQUENCE of the laws of nature.
In the next post (“Hot Car, Hot Earth”), we’ll explore the physical and chemical properties of atmospheric gasses that makes them a key player in the greenhouse effect.
—
[1] http://mediamatters.org/blog/201108090029
[2] The forecaster in question is a regular contributor to Fox News on climate matters, Joe Bastardi. According to his Wikipedia page, http://en.wikipedia.org/wiki/Joe_Bastardi, he holds a “degree in meteorology” from Penn State. He currently works for WeatherBell Analytics LLC. WeatherBell is a private company whose mission is to provide ” . . .a meteorological consulting firm that assists weather sensitive businesses to better manage their weather risk.” http://www.weatherbell.com/about-us/
[3] http://en.wikipedia.org/wiki/Greenhouse_effect





2 thoughts on “The Greenhouse Effect Part 1: Temperature and Thermodynamics”
Well done, sir. And that guy’s name really is Bastardi? Awesome.
Yes, his name is actually “Bastardi.” Now, to be fair, if we’re going to laugh at a name we should know what we’re talking about. The name is Italian, and according to some etymological information the name actually first appeared in Latin (http://www.cognomiitaliani.org/cognomi/cognomi0002as.htm). In Latin, the name means “One who is born out of wedlock.” So we are totally justified in finding this ironic. 🙂
Thanks for the positive response. I’ve written a second one on what is means to “be a greenhouse gas” and why CO2 is one. In the next post, I’ll explore how CO2+H2O = greenhouse amplification system.